Reportable events
What is reportable new information (RNI)?
During a research study, any adverse events, unanticipated problems involving risk, and non-compliance must be reported to the IRB. The guidance contained here is intended to help ensure that the reporting and review of these events occur in a timely, meaningful way so that research participants can be protected from avoidable harms. Any serious events must be reported within 24 hours. Non-serious adverse events must be reported within 5 business days. Serious events include, but are not limited to:
- death
- life-threatening event
- in-patient hospitalization
- prolongation of existing hospitalization
- a persistent or significant disability/incapacity
What must be reported?
The following information must be reported:
- protocol violation that harmed subjects or others or that indicates subjects or others might be at increased risk of harm
- information that indicates a new or increased risk, or a safety issue. For example, new information (e.g., a safety monitoring report, publication in the literature, or investigator finding) indicates an increase in the frequency or magnitude of a previously known risk, or uncovers a new risk
- complaint of a subject that indicates subjects or others might be at increased risk of harm or at risk of a new harm
- complaint of a subject that cannot be resolved by the research team
- any changes significantly affecting the conduct of the research
- any harm experienced by a research subject or other individual(s) that, in the opinion of the investigator, is unexpected and at least probably related to the research procedures
- non-compliance with the federal regulations governing human research or with the requirements or determinations of the IRB
- failure to follow the protocol due to the action or inaction of the investigator or research staff
- Breach of confidentiality
- change to the protocol taken without prior IRB review to eliminate an apparent immediate hazard to a subject
- incarceration of a subject in a study not approved by the IRB to involve prisoners
- premature suspension or termination of the research by the sponsor, investigator or institution
How do I submit a reportable event?
Report an event using the “Reportable New Information” button in ERA. The system will ask several questions and prompt you for a brief description of what happened. To report new information about a study:
- Log into http://era.oked.asu.edu/
- Click “IRB”
- Click “IRB records”
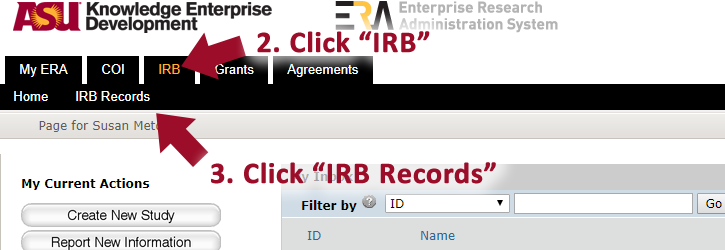
- Click “All Submissions”
- Click on the study title requiring an RNI (Reportable New Information)
- Click “Report New Information”
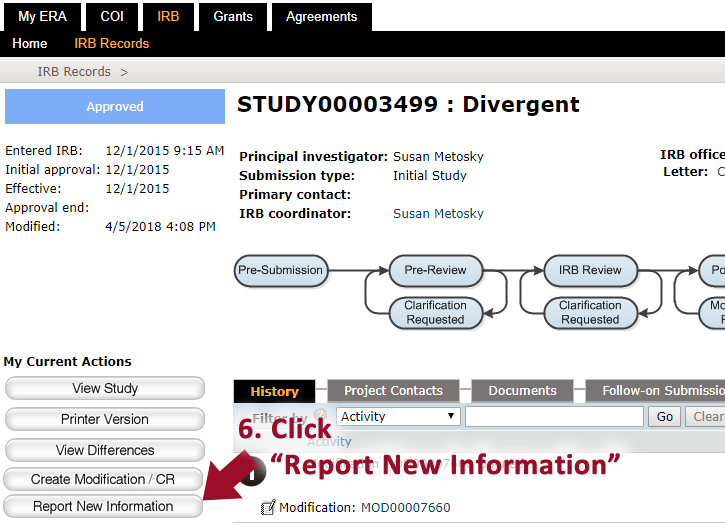
- Fill in the appropriate details. If you are unsure of what to select, contact us
- Once you are finished with all the details, click “Continue” and click “Submit RNI”
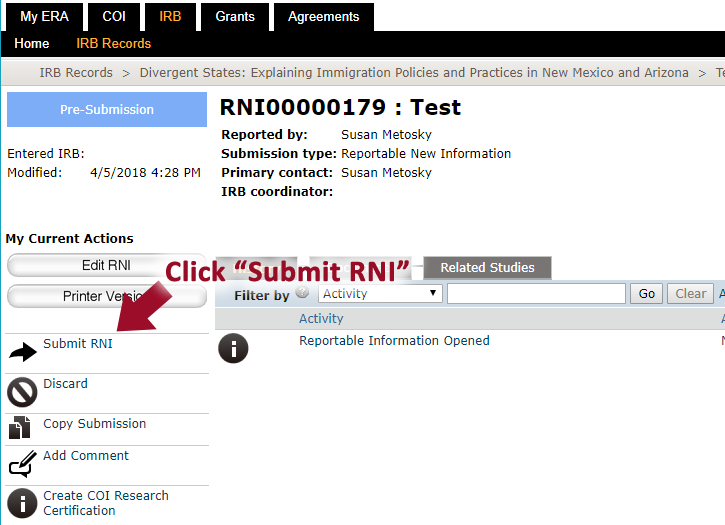
- After the RNI has been submitted, it will say “RNI Submitted” in the “History” tab.

